Current projects
We are broadly interested in understanding the functions and mechanisms of enzymes that are relevant to human health. In particular, we currently study enzymes, especially metalloenzymes, in natural product biosynthesis, bacterial and human cofactor biosynthesis, and fungal cell wall biosynthesis. For these diverse projects, we use a combination of approaches in organic chemistry, biochemistry, molecular biology, and spectroscopy (see Experimental Techniques for details).
One of our foci is the functions and mechanisms of redox-active metalloenzymes. Our primary focus is a group of enzymes called radical S-adenosylmethionine (SAM) enzymes. These enzymes form one of the largest groups of enzymes with > 700,000 functional domains. They catalyze the reductive cleavage of S-adenosyl-L-methionine (SAM) to generate transient 5’-deoxyadenosyl radical, which is subsequently used to catalyze various free radical-mediated reactions (see Figure below). Many of these enzymes are found in biological processes closely associated with human diseases. Also, these enzymes catalyze free radical-mediated reactions, which until recently, were considered rare in enzyme catalysis. Thus, their functions and mechanisms form the foundation of a novel paradigm in enzymology. We also study O2-activating non-heme Fe enzymes. Studying these two different types of enzymes gives us a unique perspective on both O2-dependent and independent radical chemistry in enzyme catalysis. See the project page for details.

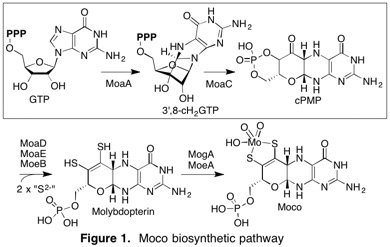
(2) Natural product and cofactor biosynthesis

Natural products biosynthesized by radical-mediated mechanisms.
(3) Antifungal antibiotics and fungal cell wall biosynthesis.

Techniques
To address our biochemical questions described above, we take diverse approaches. Each student/postdoc in the lab will learn multiple of these techniques in excellent depth and combine them to address their scientific questions. Therefore, our studies are highly interdisciplinarity in nature. The skills and knowledge obtained from these studies are translatable to many other systems, providing an outstanding training opportunity for students and postdocs. See Experimental Techniques for details.

