The detection of early carcinoma or dormant or latent metastatic tumor cells remains an elusive but important clinical goal. We seek to develop further revolutionary new nanotechnology that enables optically based detection of metastatic cancer cells. Towards this goal, we will further refine design criteria for near infrared (NIR) emissive polymersomes, a promising new soft matter nanoscale platform for in vivo diagnostic and drug-delivery applications.
This program will develop (i) targeted nanoscale (diameter <= 100 nm) NIR-emissive polymersomes, (ii) nanoscale NIR-emissive polymersomes having optimized emissive output, (iii) prototype targeted nanoscale vesicles in which the polymeric building blocks are based-upon FDA-approved materials, (iv) targeted NIR-emissive polymersomes with ideal cell-surface adhesion dynamics, and (v) methods and technology that provide not only new insights into metastatic disease, but define an evolvable nanoscale platform for in vivo dormant tumor cell detection, diagnosis, and treatment.
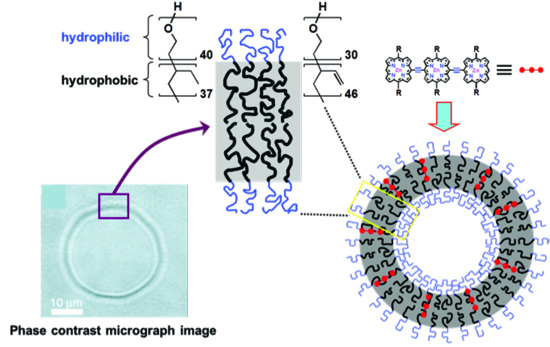
These efforts involve correlating NIR fluorophore structure and photophysics, polymersome composition of matter, vesicle mechanical and biological properties, nanoscale NIR-emissive polymersome fluorescence output, and the nature of the cellular targeting motif, with in vivo function and efficacy. As such, the experimental approach we pursue is cross-cutting and integrative, encompassing supramolecular chemical synthesis, photophysical characterization, in vivo imaging, bioengineering, biology, and medicine. We strive to establish design principles that will ultimately enable real-time detection and identification of limited target cell numbers under clinically relevant diagnostic conditions, and define new tools for the study of metastatic disease.