The Chen Lab is interested in understanding the molecular and genetic events underlying cancer progression and metastasis. The focus of our work is a series of genetically engineered mouse models that faithfully recapitulate human disease. We have generated mouse models of several cancer types, including prostate cancer and lung cancer, as well as derived organoid cultures from mutant mice to study the biological functions of cancer related genes. Using a multidisciplinary approach combining mouse genetics, omics technologies, cross-species analyses and in vitro approaches, we aim to identify cancer cell–intrinsic and –extrinsic mechanisms driving aggressive and metastatic disease, with a long–term goal of developing new therapeutic strategies for preventing and treating human cancer. Research in the Chen Lab mainly focuses on five areas:
(1) Mechanisms of Metastasis
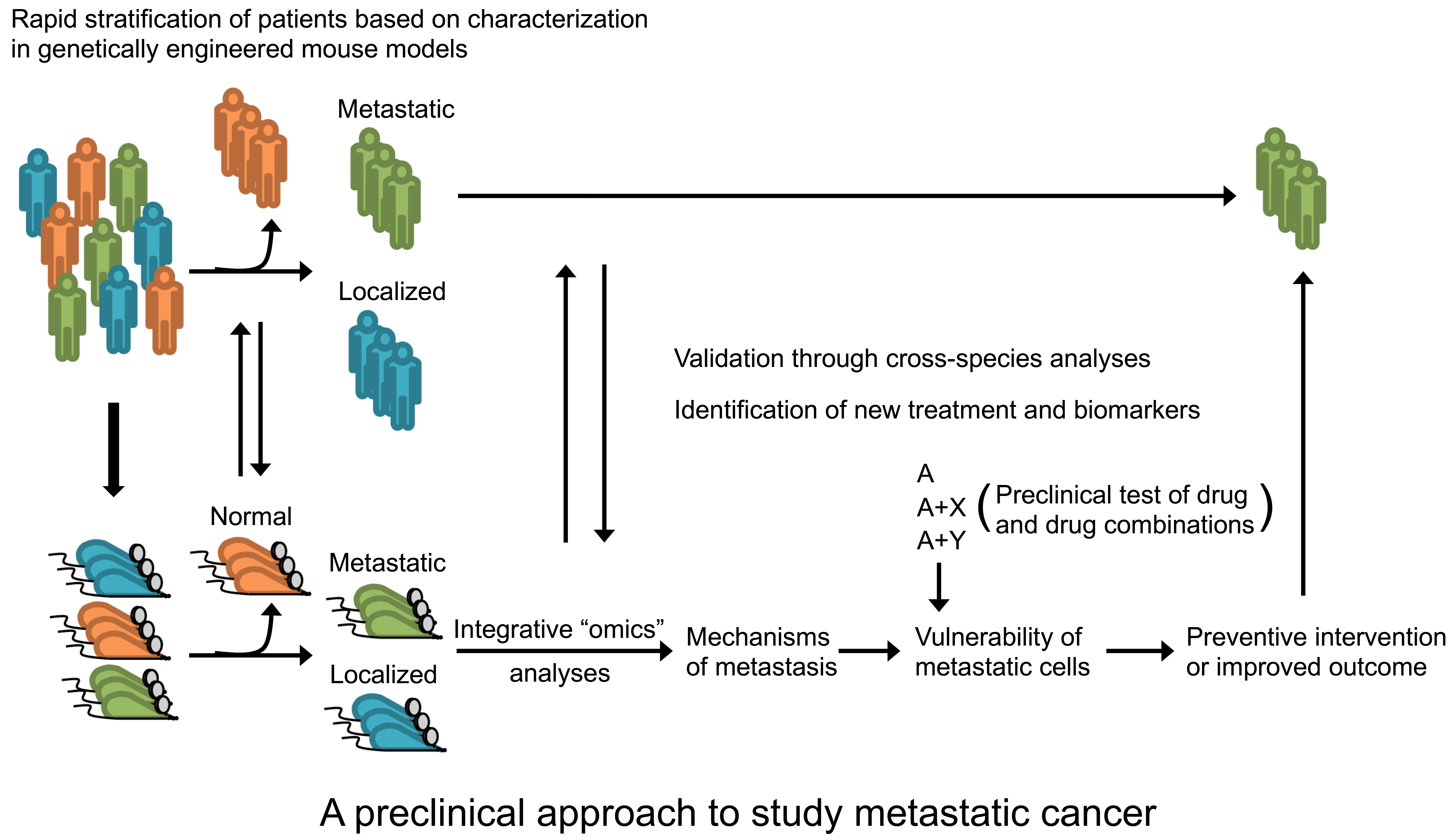
Metastasis accounts for more than 90% of cancer-related deaths and yet remains one of the least understood aspects in cancer biology. One objective in our research is to identify mechanisms of metastasis and vulnerability of metastatic cells. We are among a few groups that generate mouse models of metastatic prostate cancer based on prostate epithelium-specific inactivation of Pten (Nature Genetics 2018). By performing integrative omics analyses of tumors from localized and metastatic mouse models, we aim to reveal transcriptional, translational and metabolic mechanisms that drive metastatic disease. Additionally, by conducting preclinical studies of drugs targeting vulnerability of metastatic cells, we hope to find new strategies for preventing or slowing the spread of cancer in mice, and ultimately translating key findings into the clinic to treat metastatic disease (Cold Spring Harbor Perspectives in Medicine 2018).
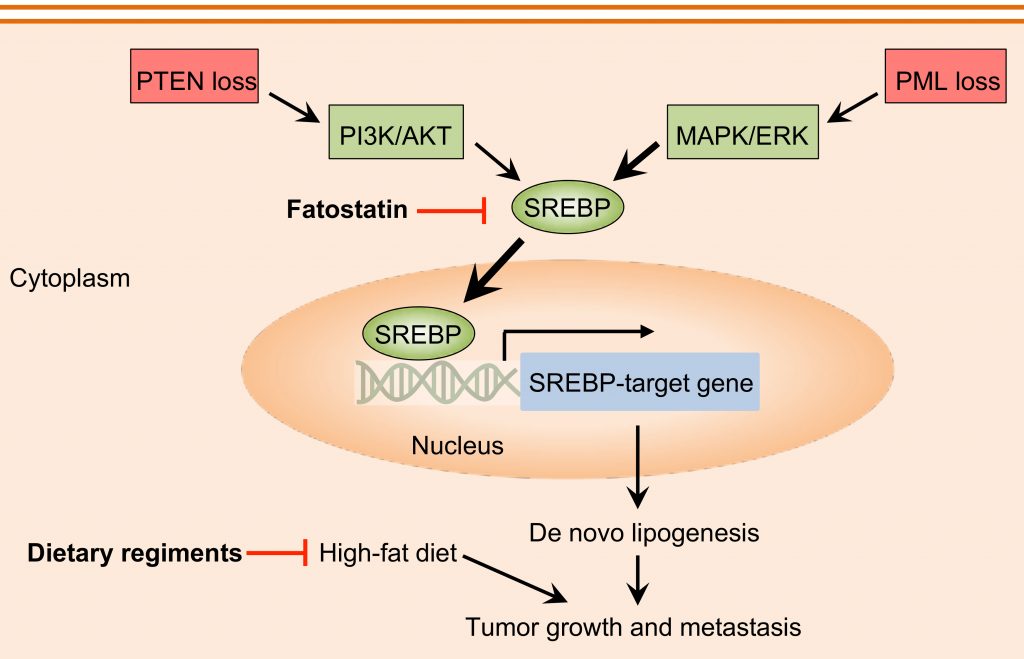
(2) Lipid Metabolism and Cancer
Cancer cells undergo metabolic reprogramming to support cell proliferation, growth and dissemination. Alterations in lipid metabolism, and specifically the uptake and synthesis of fatty acids, comprise one well-documented aspect of this reprogramming (Precision Clinical Medicine 2019). Our recent studies suggest that co-activation of the PI3K/AKT and MAPK pathways converges their oncogenic functions through SREBP-dependent lipogenic program, and that lipids obtained through de novo lipogenesis or dietary uptake may provide a cell-intrinsic signal promoting the aggressiveness of cancer (Nature Genetics 2018). A current focus in the lab is to identify key genes and pathways that mediate lipid-driven tumorigenesis and to establish the molecular mechanisms connecting obesity and cancer aggressiveness.
(3) Ferroptosis as a Therapeutic Target in Cancer
Ferroptosis is a form of regulated cell death driven by iron-dependent lipid peroxidation, especially of phospholipids containing polyunsaturated fatty acid at the plasma and organellar membranes. It has been highly conserved from protozoa, plants, and fungi to metazoans, and represents an ancient vulnerability that could be harnessed for cancer therapy. Leveraging recent insights from our research, we have determined that advanced prostate cancer is highly sensitive to the induction of ferroptosis, especially in the context of loss of the RB1 tumor suppressor gene (Journal of Clinical Investigation 2023). We are currently exploring whether targeting ferroptosis, either through single or combination agents, could represent an effective therapeutic approach to treating lethal prostate cancer.

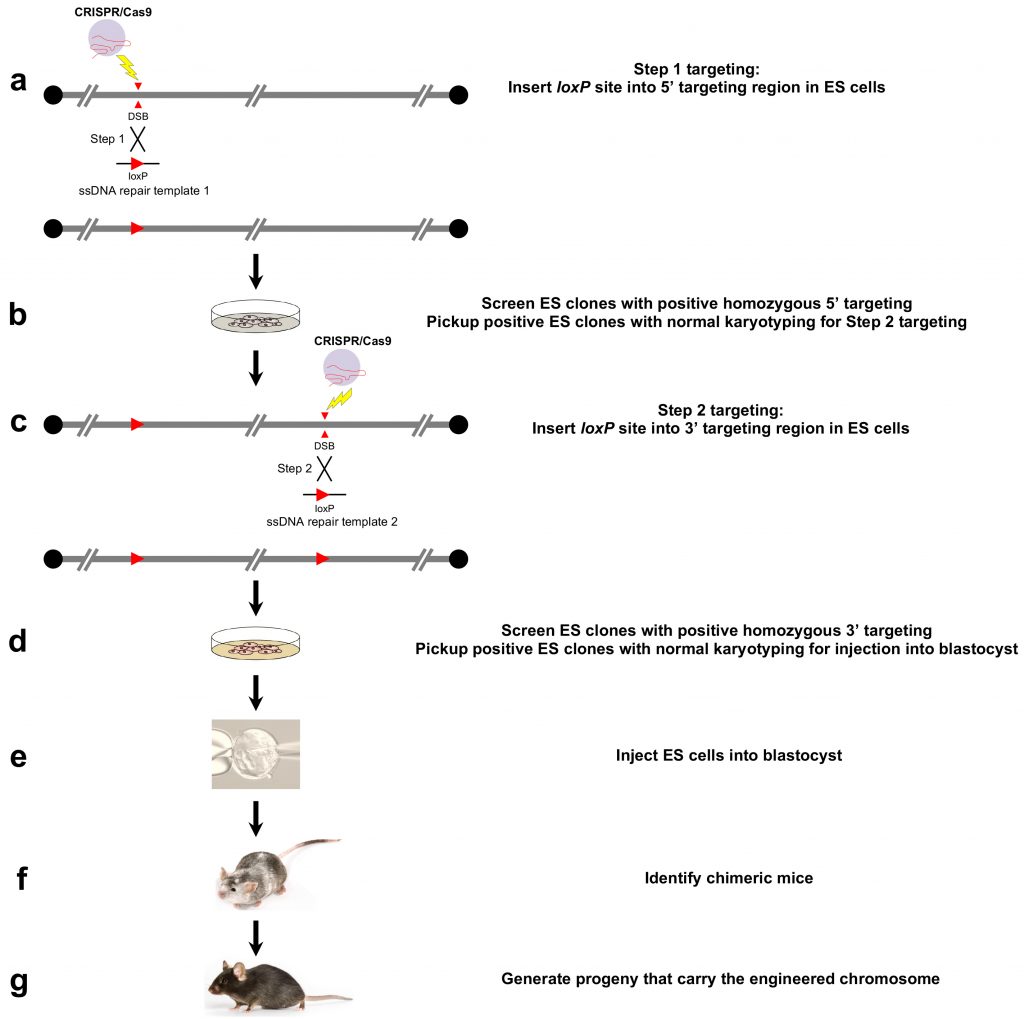
(4) The Etiology of Large Chromosomal Deletions in Human Cancer
Little is known about the biological and phenotypic impacts of large chromosomal deletions in cancer despite their occurrence 30 times more frequently than focal deletions (Nature 2010). Due to the limited cloning capacity of targeting vectors (~300 Kb) and the rarity of on-target homologous recombination events in traditional gene-targeting technology, modelling large chromosomal deletions in mice has proved highly challenging, and the in vivo role of such lesions in tumorigenesis has therefore been significantly understudied. To fill this critical gap, we have developed an innovative approach to chromosomal engineering over large genetic distances through CRISPR/Cas9 technology in mouse embryonic stem (ES) cells. The resulting ES clones carrying the desired genome edits make possible the creation of conditional knockout mice that accurately mimic cancer-associated large deletions. We are currently utilizing our innovative approach to generate and fully characterize novel GEM models that harbor highly recurrent large chromosomal deletions in human cancer, with a goal of validating and credentialing these models as genetically and biologically robust representations of human cancer.
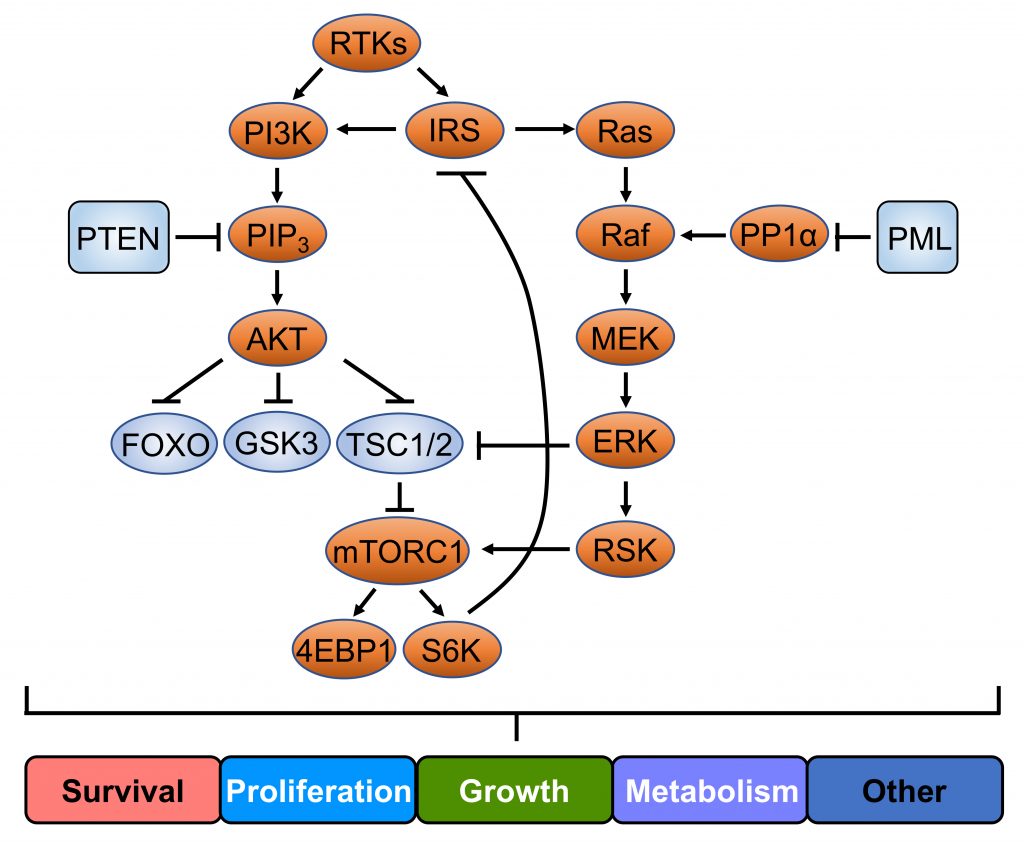
(5) PTEN/PI3K/AKT and MAPK Signaling Pathways
PTEN/PI3K/AKT and MAPK signaling pathways govern a variety of biological processes, including cell survival, proliferation, growth and metabolism (Nature Reviews. Molecular Cell Biology 2018; Nature Communications 2018). They are the most commonly altered pathways in human cancer and their cross-talk is also of key importance in the regulation of tumorigenesis. We have recently revealed a PP1α-PML molecular network that is genetically altered in human cancer towards aberrant MAPK activation in PTEN loss/PI3K-AKT driven cancers (Nature Communications 2018). Our studies are now focused on developing optimal combinatorial therapies that target different components of the pathway to combat human cancer.