Acetaminophen
Acetaminophen (N-acetyl-para-aminophenol, APAP or paracetamol) is the most widely used over-the- counter and prescription painkiller in the world. While safe at therapeuticdoses of up to 4 grams per day for adults, acetaminophen overdoses, either accidental or intentional, are the leading cause of acute liver failure in the United States, accounting for some 56,000 emergency room visits, 2,600 hospitalizations and nearly 500 deaths annually.
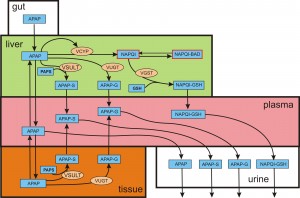
Acetaminophen is metabolized in the liver by conjugation with sulfate and glucoronidate. The conjugates are inert and are excreted in the urine. Depending on dose, a fraction of APAP is converted into a highly reactive toxic intermediate, N-acetyl-p-benzoquinone imine (NAPQI) by several P450 cytochromes. Substantial amounts of NAPQI are effectively eliminated by conjugation with glutathione (GSH). However, after a large dose of APAP, the sulfonation reaction becomes saturated and the build up of NAPQI depletes GSH in the liver, causing further accumulation of NAPQI. Unconjugated NAPQI binds to proteins and subcellular structures and induces rapid cell death and necrosis that can lead to liver failure. The main biochemical pathways of acetaminophen metabolism and the transports between various compartments are pictured in the figure below.
Whole body metabolism of acetaminophen (click figure)
Glutathione (GSH) is a tri-peptide made of cysteine, glutamate, and glycine. Of the three, cysteine has by far the lowest normal concentration in liver cells. Therefore, N-acetylcysteine (NAC) is an effective antidote for APAP poisoning because it provides extra cysteines for GSH synthesis in the liver. Current protocols recommend treating patients with an initial dose of 150 mg/kg NAC, infused over a period of an hour, upon hospitalization, followed by decreasing amounts of NAC infused over the next 20 hours. Fatal liver damage can usually be prevented if the initial dose of NAC is administered within 8-12 hours of an APAP overdose. This antidote dosage regime has been developed empirically over a period of many years based on outcomes from clinical cases. It is not known whether the current NAC treatment protocol is optimal.
In 2013, we developed a mathematical model corresponding to the above diagram (Ben-Shachar et al. 2013), including the liver biochemistry of APAP metabolism. Rotem Ben-Shachar was first author. A previous paper (Remien et al., Heptology 56(2012), 727-734) had developed a mathematical model of liver enzymes in the plasma and cell necrosis in the liver in order to predict which patients require liver transplants. Our paper used the Remien model of cell death in the liver. We showed that our model accurately reproduces published clinical and experimental data on the dose-dependent time course of acetaminophen in the plasma, the accumulation of acetaminophen and its metabolites in the urine, and the depletion of glutathione caused by conjugation with the toxic product. We used the model to study the extent of liver damage caused by overdoses or by chronic use of therapeutic doses, and the effects of polymorphisms in glucoronidation enzymes. We used the model to study the depletion of glutathione and the effect of the size and timing of N-acetyl-cysteine doses given as an antidote. Our model accurately predicts patient death or recovery depending on size of APAP overdose and time of treatment. Our model uses a previously developed model of hepatic GSH metabolism (Reed et al., 2008).
The regeneration of GSH after an APAP dose is also affected by the concentration of glutamate in the liver and it is known that supplementation of rats with glutamine prior to and APAP over dose acts to preserve GSH levels. This suggests that in the presence of APAP overdose the concentration of glutamate may also be rate-limiting for the regeneration of GSH. The metabolism of glutamate and glutamine in the body is quite complicated because glutamine is produced from glutamate in muscles. To understand these phenomena we developed a whole body model of glutamate and glutamine metabolism (Bilinsky et al. 2015). Lydia Bilinsky was the first author on the paper. We show that the model reproduces precisely the empirically observed decline in liver GSH during starvation, as well as its diurnal variation under normal fed conditions. We use the model to investigate changes resulting from a 9-day course of dexamethasone treatment. And, we use the model to study APAP overdose and investigate the mechanism by which glutamine preserves hepatic GSH levels.
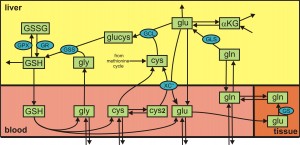 Whole body glutamate and glutamine metabolism (click image)
Whole body glutamate and glutamine metabolism (click image)
In the course of this second study, it came to our attention that there are large species and gender differences in the metabolism of APAP in the liver. These differences are currently under study.