Neurotransmitters provide the mechanism by which electrical signals are communicated from one neuron to the next. However, as we discussed in the tab “Volume Transmission,” there is strong evidence that in many cases it is the concentration of a neurotransmitter in the extracellular space of a nucleus that affects electrophysiological neurotransmission by other neurotransmitters. This raises several natural questions. What are the mechanisms by which the extracellular concentrations of neurotransmitters are controlled? How do neurotransmitters in the extracellular space affect synaptic transmission by other neurotransmitters? How robust are these mechanisms in the face of polymorphisms in the enzymes affecting synthesis, release, and reuptake of neurotransmitters? How are dysfunctions in these control mechanisms related to neurological and neuropsychiatric diseases? In this section we briefly describe our work on several of these questions.
Passive stabilization of DA in the striatum. Parkinson’s disease is a neurodegenerative disorder associated with cell loss from the substantia nigra pars compacta (SNc). The dopaminergic cells of the SNc send projections to the striatum where the loss of dopaminergic tone is thought to be the main cause of tremor and other motor symptoms of Parkinsonism. An interesting and important feature of the disease is that symptoms do not appear until a very large percentage (75%-90%) of SNc cells have died and therefore this feature has been the focus of much experimental and clinical investigation. Experiments with animal models [6e8] have shown that although striatal tissue content of dopamine declines more or less proportionally to cell death in the SNc, the extracellular concentration of dopamine (EDA) in the striatum remains near normal until more than 85% of SNc neurons have died. This is widely believed to be the reason that symptoms do not appear until very late in the degeneration of the SNc.
What is the basis of this remarkable homeostasis of striatal EDA in the face of progressive cell death in the SNc? Some researchers proposed that the nigrostriatal system adapts to cell death to maintain extracellular DA level by increasing DA synthesis in the living terminals or by sprouting more terminals. But in 2003 Bergstrom and Garris (J. Neurochem 87,1224-36) proposed a very simple explanation that they called “passive stabilization’’ and provided experimental evidence for it. The extracellular concentration of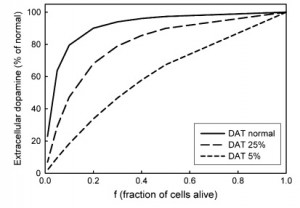 DA depends on the balance between release of DA and reuptake of DA by the dopamine transporters (DATS). If half of the SNc cells die, there will be only half as much release, but there will also be only half as much reuptake, so the concentration of DA in the extracellular space should remain the same.
DA depends on the balance between release of DA and reuptake of DA by the dopamine transporters (DATS). If half of the SNc cells die, there will be only half as much release, but there will also be only half as much reuptake, so the concentration of DA in the extracellular space should remain the same.
We used our mathematical model of a DA terminal (Best et al. 2009) to investigate the proposal of Bergstrom and Garris (Reed et al 2009). Notice that their hypothesis does not explain why passive stabilization breaks down when, f, the fraction of SNc cells left alive gets small. We believe that passive stabilization breaks down at small f because there is always some removal of DA from the system in the extracellular space by uptake into glial cells and blood vessels or simply diffusion out of the tissue. As the number of DA terminals in the striatum gets smaller, these removal effects get proportionally larger because the reuptake DATs become sparser and sparser. This hypothesis was confirmed and explained by our mathematical modeling. The figure above shows the concentration of DA in the striatum in the model as a function of f, the fraction of SNc cells left alive. One can see that the passive stabilization effect of Bergstrom and Garris keeps the extracellular DA concentration quite constant until approximately 80% of the SNc cells have died. As even more cells die the concentration drops to zero because the removal effects dominate more and more. The dashed curves show that the passive stabilization depends on the dopamine transporters.
Control of extracellular DA by autoreceptors. Under the tab “constructing models’’ we gave as an example our 2009 model (Best et al. 2009) of a DA terminal including synthesis, packaging into vesicles, release, and reuptake via the DATs. We also included the effects of the DA autoreceptors that sense the DA concentration, EDA, in the extracellular space. When DA gets higher than normal, the autoreceptors inhibit synthesis and release of DA, and when DA gets lower than normal this inhibition is withdrawn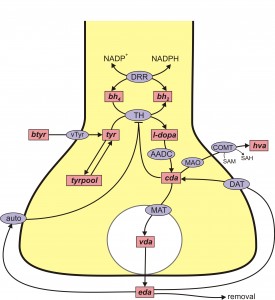 stimulating synthesis and release. Thus the autoreceptors act to modulate EDA against both long term and short term perturbations such as changes in the supply of tyrosine or changes in firing rate. The mechanisms by which EDA affects synthesis and release via the autoreceptors are mostly unknown and an important topic of current research that involves difficult questions in cell biology. The control of DA in the extracellular space is also affected by other neurotransmitters. For example, there is dense serotonergic projection to the striatum from the dorsal raphe nucleus (DRN). The released 5HT binds to 5HT receptors on DA terminals and increases DA release when the SNc cells fire.
stimulating synthesis and release. Thus the autoreceptors act to modulate EDA against both long term and short term perturbations such as changes in the supply of tyrosine or changes in firing rate. The mechanisms by which EDA affects synthesis and release via the autoreceptors are mostly unknown and an important topic of current research that involves difficult questions in cell biology. The control of DA in the extracellular space is also affected by other neurotransmitters. For example, there is dense serotonergic projection to the striatum from the dorsal raphe nucleus (DRN). The released 5HT binds to 5HT receptors on DA terminals and increases DA release when the SNc cells fire.
Homeostasis of DA and cryptic genetic variation. An important field of study in the past 15 years has been to quantify the effects of gene polymorphisms on the proteins that are important for the dopaminergic system, for example, tyrosine hydroxylase (TH) or the dopamine transporter (DAT). Typically, these experiments are done in test tubes or in vitro and the polymorphisms often have large quantitative effects on the function of the proteins. And, it is very tempting to conclude that the polymorphisms are therefore the “causes’’ of various neurological or neuropsychiatric diseases. However, in vivo there are many control mechanisms (we’ve discussed two above) that buffer the DA concentration in the extracellular space against perturbations in the DA system. We pointed this out already in our 2009 paper (Best et al. 2009), but the point is made dramatically by the two dimensional surface taken from our 2014 paper (Nijhout et al. 2014). The surface shows the extracellular DA concentration (z-axis) at steady state as a percentage of normal as a function of the activity of tyrosine hydroxylase and the efficacy of the dopamine transporter computed from our model. In both cases, 1 indicates normal activity for TH and DAT. The large white dot on the surface is wild type, the concentration of EDA when TH and DAT have their normal activities. The smaller white dots on the surface indicate points that correspond to common polymorphisms (homozygotes and heterozygotes) in the human population taken from the table. Notice that all the white dots lie on the flat part of the surface where the polymorphisms cause only very modest changes in extracellular DA despite the fact that they cause large changes in protein activity. This is the effect of the autoreceptors. It is quite amazing that all these polymorphisms all lie on the flat part of the surface. Presumably, if they didn’t, they would have been selected against and would not be common in the human population. This example shows why one has to be very careful about jumping to physiological conclusions from test tube or in vitro experiments.
This surface is a perfect example of cryptic genetic variation in which large variation in genes (gene products), that is, TH and DAT, produce very little variation in a phenotypic variable, the extracellular DA concentration. It should be kept in mind that the actual situation is much more complicated than this two dimensional surface in three space would lead one to believe. There are many other variables, both genetic variables (for example a polymorphism in the monoamine transporter) or phenotypic variables (for example the 5HT concentration, see below) to could affect the shape of this surface. The “real’’ surface is a high dimensional surface in a high dimensional space. Nevertheless this surface does tell us a lot, and it is interesting to think about the people who have the 15% mutation in TH. They are the ones to the right sitting at the edge of the cliff where DA drops to zero. Interestingly, these genotypes sometimes show a dystonia, involuntary muscle contractions that affect posture, brought about by low levels of extracellular DA that can be alleviated by levodopa. So, one could say that their position at the edge of the cliff (that is, having the 15% TH polymorphism) predisposes them to the dystonia. Some of them are pushed over the cliff by other variables not pictured and thus show the dystonia. The job of a precision medicine provider would be to advise a patient with the 15% TH polymorphism how to flatten the region around where they lie on the surface and thus to avoid being pushed over the cliff by other variables. In our simulations, the region around these individuals gets flatter if one increases the strength of the autoreceptor effect.
Does 5HT stabilize DA in the striatum? The dopaminergic cells in the SNc project to the striatum where they stimulate spiny neurons in direct pathway from the Cortex to the thalamus and they inhibit spiny neurons in the indirect pathway. The 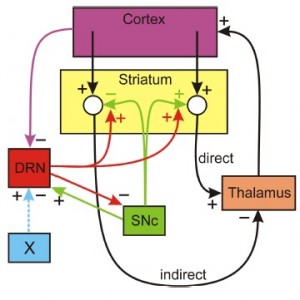 direct pathway stimulates the thalamus and the indirect pathway inhibits the thalamus. The simplest conceptual model of Parkinson’s disease is that when SNc cells die the extracellular DA concentration drops in the striatum removing excitation from the direct pathway and removing inhibition from indirect pathway, thus tipping the balance towards the indirect pathway that inhibits the thalamus. The resulting inhibition of the thalamus cause the inability to initiate movement. But, what does this have to do with 5HT? There is a dense 5HT projection to the striatum from the dorsal raphe nucleus (DRN) and it is known that increased concentrations of 5-HT in the striatum facilitate the release of DA from the dopaminergic projections from the SNc. Thus 5HT could partially compensate for cell loss in the SNc if the DRN fires more when SNc cells die. There is a plausible mechanism by which this could happen. When SNc cells die there is more inhibition of the thalamus (as discussed above). Since projections from the thalamus excite cortical neurons there will be less stimulation of the cortex. There are numerous projections from higher brain regions to the DRN; in particular, there are inhibitory projections from medial prefrontal cortex and if these projections fire less, then the DRN neurons would fire more. This is speculative, but gives a plausible mechanism by which the 5HT projection from the DRN to the striatum acts to stabilize DA in the striatum. That this idea would work is supported by a simple mathematical model (Reed et al. 2013), but not enough is known about the projections from the thalamus to the cortex and from the cortex to the DRN to be sure of the anatomy.
direct pathway stimulates the thalamus and the indirect pathway inhibits the thalamus. The simplest conceptual model of Parkinson’s disease is that when SNc cells die the extracellular DA concentration drops in the striatum removing excitation from the direct pathway and removing inhibition from indirect pathway, thus tipping the balance towards the indirect pathway that inhibits the thalamus. The resulting inhibition of the thalamus cause the inability to initiate movement. But, what does this have to do with 5HT? There is a dense 5HT projection to the striatum from the dorsal raphe nucleus (DRN) and it is known that increased concentrations of 5-HT in the striatum facilitate the release of DA from the dopaminergic projections from the SNc. Thus 5HT could partially compensate for cell loss in the SNc if the DRN fires more when SNc cells die. There is a plausible mechanism by which this could happen. When SNc cells die there is more inhibition of the thalamus (as discussed above). Since projections from the thalamus excite cortical neurons there will be less stimulation of the cortex. There are numerous projections from higher brain regions to the DRN; in particular, there are inhibitory projections from medial prefrontal cortex and if these projections fire less, then the DRN neurons would fire more. This is speculative, but gives a plausible mechanism by which the 5HT projection from the DRN to the striatum acts to stabilize DA in the striatum. That this idea would work is supported by a simple mathematical model (Reed et al. 2013), but not enough is known about the projections from the thalamus to the cortex and from the cortex to the DRN to be sure of the anatomy.