The Schumacher Lab has determined the molecular mechanisms of several important
biological systems. The main focus of the lab is the dissection of essential mechanisms
involving protein-nucleic acid interactions, in particular as they relate to microbial
pathogenesis.
Our Experimental Approach
We employ X-ray crystallography, cryo-electron microscopy and a wide range of
biochemical and biophysical methodologies as well as some cellular techniques.
Research in Transcription Regulation
Our studies on transcription regulation largely focus on global transcription regulatory
networks that control entire developmental processes.
Selected contributions to understanding transcription processes.
- The modern field of regulatory biology was founded on studies of the Lac repressor,
but its mechanism of DNA binding had remained unknown. We obtained the first
structure of a LacI protein, PurR, bound to DNA, unveiling the general DNA binding
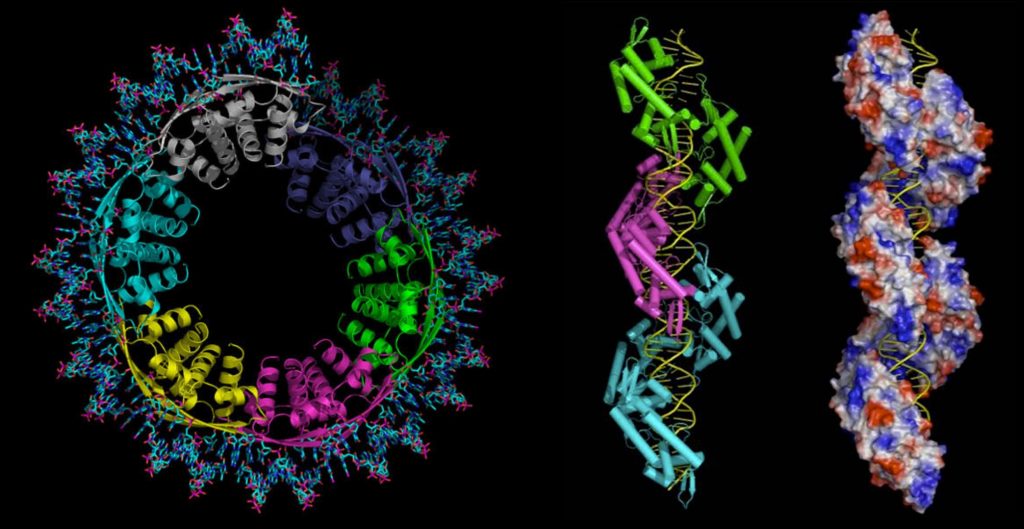
mechanism for LacI members. - We determined the molecular transcriptional mechanisms controlling nitrogen and
carbon homeostasis in the model Gram-positive bacteria B. subtilis, including complex
formation of CcpA-phosphoserine-HPr-DNA and structures of all components of the
nitrogen circuitry composed of TnrA-DNA, GlnR-DNA. We revealed these transcription
factors are themselves regulated by the central enzyme of nitrogen control, GS,
whereby GS binding to TnrA and GlnR causes an unprecedented dodecamer to
tetradecamer transition that we are now studying by cryo-EM. - Unlike other eukaryotes, more than 80% of the promoters of the pathogen, T.
vaginalis, are controlled by its DNA binding, Inr binding protein (IBP). We obtained the
structure of the T. vaginalis IBP-DNA complex and discovered that the IBP directly
contacts and recruits the C-tail of T. vaginalis RNAP, leading to a mechanism of
activation. - In collaboration with Dr. Mark Buttner’s lab at the John Innes Centre we are
delineating the steps that control the development and antibiotic (secondary metabolite)
production in Streptomyces, our main source of clinically important antibiotics. These
studies uncovered c-di-GMP as a key signaling molecule, and we showed that a novel
tetrameric form of c-di–GMP binds directly to the master developmental BldD. - We are performing cryo-EM analyses on RNAP bound to promoter DNA and DNAbinding
regulatory proteins, and providing atomic level insight into how the DNA binding
activators coordinate directly with RNAP.
Research in kRNA Editing
One part of my laboratory focuses on a truly bizarre form of RNA editing, which takes place in the mitochondria of kinetoplastid parasitic protozoa such as T. brucei, called kinetoplastid RNA editing (kRNA editing). In extreme forms of kRNA editing, hundreds of uridines (Us) are inserted or deleted to generate translatable mRNAs, as dictated by gRNAs. We focus on the functions and structures of unique RNA editing accessory factors that are required for this unusual process. We solved the MRP1/MRP2 complex, which we showed functions as an RNA matchmaker between gRNA and pre-edited mRNAs. Highly edited transcripts have multiple G-rich stretches, which must be melted to allow for the insertion and removal of Us. We recently obtained structures of what is arguably the most important accessory factor, TbRGG2, and showed it binds specifically to poly-G and poly-U substrates and melts the RNA secondary structure suggesting it may be an important modulator of RNA structure in kRNA editing.
DNA Segregation
A major research focus of the lab is DNA segregation or partition—one of the most fundamental processes in biology. We mainly study segregation of low copy number prokaryotic plasmids, which are a main contributor to the rise in multidrug resistance. Bacterial partition (par) systems also require only 3 components, making them ideal model systems for atomic level study. These three elements encoded on the plasmid, include a DNA centromere, centromere-binding protein and an NTPase. In the last 15 years we have determined the first structures of partition complexes and delineated mechanisms for all three types of systems. These studies show these systems use 1) actin like polymers, 2) tubulin like polymers and 3) walker box proteins, with a monomer-to dimer switch controlling association with the host nucleoid, hence a piggy-back like mechanism. Recently, we showed walker box systems are widespread in archaea, making them arguably the most common type of segregation system in nature. Moreover, we have structurally characterized walker box like proteins used to transport bacterial organelles, indicating this as a universal macromolecular transport system.
Coordination of Segregation With Cell Division and Replication
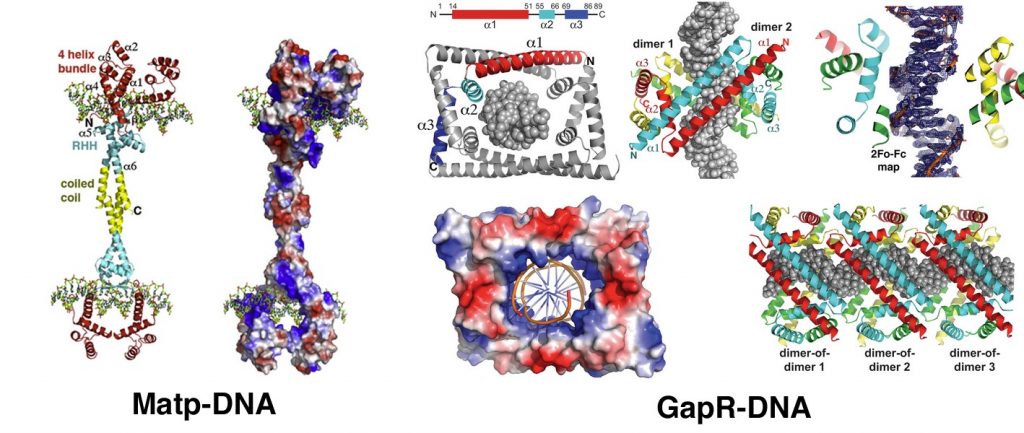
In addition to the process of DNA segregation we also interrogate how chromosomal DNA segregation in prokaryotes is coordinated with replication and cell division. Key findings include the molecular mechanisms by which the nucleoid occlusion protein SlmA binds specific chromosomal sites as well as FtsZ to prevent septum formation through the E. coli chromosome. We also obtained a structure of MatP-DNA that showed how the Ter specific protein may act to positively recruit FtsZ to the cell center. In Caulobacter, we obtained the structure of the NAP GapR bound to AT rich DNA, revealing how a protein can recognize a DNA conformation to promote topoisomerase activity.

Mechanisms of Multidrug Recognition and Multidrug Tolerance
As a postdoc, Dr. Schumacher addressed a key question in the field of multidrug resistance (MDR), which is how a single protein can recognize multiple, structurally dissimilar compounds. The lab has followed up with studies on the phenomenon of Multidrug Tolerance (MDT) in collaboration with the Brennan lab. We characterized the first identified MDT system, that of the E. coli HipA/HipB. These studies provided the first mechanistic insights into heritable, clinically relevant multidrug tolerance.