Cocaine, like many drugs1 can enter the body a variety of ways (Figure 4).
Figure 4 Modes of drug administration into the body. Red is the arterial side and blue is the venous side. Adapted from Ray O. and Ksir C. (2002). Drugs, Society, and Human Behavior, pg. 154. McGraw-Hill: New York.
The easiest way to get cocaine or any drug into the bloodstream is to inject it directly into a vein since there are no membranes to traverse in order to get there. In contrast, if cocaine is smoked, snorted2 or ingested by mouth, it must pass several membrane barriers by diffusion before reaching the bloodstream, and some of these processes are slower than others. Smoking a drug such as cocaine, nicotine or heroin, enables its diffusion into the bloodstream almost as fast as injecting (also see Figure 5). The lungs have a very large surface area for absorption of the drug from the alveoli (very small sacs where gas is exchanged) into the capillaries. Capillaries are the smallest form of blood vessels and are very numerous. In fact they are able to deliver nutrients such as oxygen and glucose to every cell in the body. They also pick up waste such as carbon dioxide and metabolic products. Once in the capillaries, the drug travels in the oxygenated blood to the heart and then gets pumped throughout the body (organs such as brain, liver and kidneys that have a high blood flow receive blood first) to reach all cells (Figure 4).
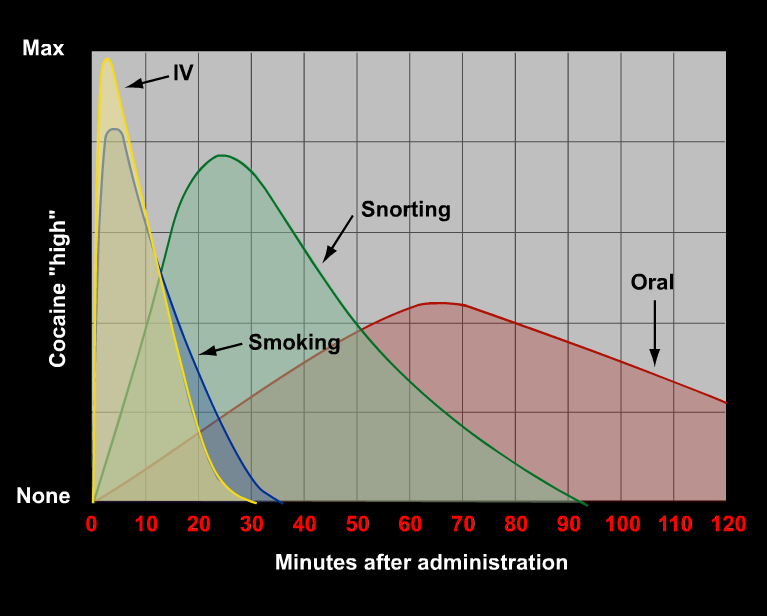
Figure 5 The relationship between the mode of cocaine intake and the intensity and duration of its euphoric effects.
However, the brain only permits the entry of certain kinds of drugs by diffusion; only those drugs that exist in their unionized form are able to diffuse through the group of membranes that form the “blood brain barrier3“. The blood brain barrier consists of tightly packed capillary endothelial cells4, so there are no pores through which charged compounds can pass (Figure 6). Drugs that are highly lipophilic5 (lipid-loving) such as cocaine penetrate most quickly by diffusion. Other drugs that cross the blood brain barrier easily include nicotine, marijuana and heroin. (Some larger and ionized drugs can be transported across the blood brain barrier by binding to transport proteins.) By restricting only certain molecules (and drugs) from reaching the brain, the brain can be protected from dangerous compounds. If a drug such as cocaine is snorted, it takes a little longer to get to the brain where it produces its psychoactive6 effects. Cocaine diffuses through special epithelial cells7 (mucosal cells8) lining the nasal passages into nearby capillaries. There is a very extensive capillary network in the nasal passages so drugs are absorbed into the bloodstream fairly quickly. Then, the cocaine travels within the venous system to the heart, then to the lungs, and back to the heart for distribution throughout the body (Figure 4).
Figure 6 Left: A cross section of a non-brain capillary showing that both lipophilic or non-polar (unionized) and polar (ionized) molecules can pass through spaces between endothelial cells. Right: In brain capillaries, the endothelial cells are tightly packed and so there is a barrier to prevent polar (ionized) molecules from diffusing into the brain. Only lipophilic or non-polar molecules can diffuse through the endothelial cell membranes.
The slowest way to get a drug into the bloodstream is by ingestion. When a drug is ingested by mouth, it travels first to the gut, where it must pass through mucosal cells lining the gut in order to get to the capillaries. Here in the gut, the pH is an important determinant of the drug’s chemical charge, and thus its ability to pass through the mucosal cell membranes. Most drugs are weak acids9 or bases and their charge will be determined by the pH of the surrounding medium. The very acidic pH of the stomach will keep weak acids in their uncharged (non-polar10 or unionized) form. In contrast, weak bases11, like cocaine, will be mostly charged or ionized12 (polar)13 at low pH. As discussed above, ionized compounds have difficulty passing through most membranes (other than capillaries), while unionized compounds are more lipophilic, and slide through the membrane easily. Another factor that slows the appearance of drugs taken by mouth into the brain is metabolism. The capillaries in the gut connect to blood vessels that go directly to the liver, so as drugs leave the gut they travel to the liver first (this is called the portal circulation). There, some of the drug is metabolized and inactivated as it passes through. Metabolites are often more polar forms of the parent drug, allowing them to be excreted in the urine more easily. After the drug leaves the liver, it travels to the heart, then to the lungs and finally back to the heart to be distributed throughout the rest of the body (Figure 4). Thus, in this case, less cocaine (in its active form) reaches the brain. This is one reason why abusers do not take cocaine orally (see below).
Definitions:
1 A substance that affects the structure or function of a cell or organism.
2 To breathe in a compound in solid form through the nostrils. With reference to cocaine, it is the hydrochloride salt.
3 A tightly joined layer of cells lining the capillaries in the brain. It restricts passage of drugs and other molecules across the cell layer into the brain to include only those that are lipophilic (uncharged).
4 Cells that line the blood vessels and capillaries. Unlike epithelial cells, these cells have no contact with the environment outside the body.
5 High lipid solubility. Lipophilic compounds dissolve readily in oil or organic solvent. They exist in an uncharged or non-polar form and cross biological membranes very easily.
6 Pertains to drugs that act in the brain to produce changes in mood, perceptions, and behavior.
7 Cells that line all free surfaces such as the skin, nasal passages, and the gut.
8 A special type of epithelial cell that can secrete mucous. This protects the cells lining the tissue.
9 A compound that tends to give up a H+ when placed in an alkaline solution.
10 A chemical property of a substance that indicates an even distribution of charge within the molecule. A non-polar or non-charged compound mixes well with organic solvents and lipids but not with water.
11 A compound that tends to accept a H+ when placed in an acidic solution.
12 An atom, radical, or molecule that has gained or lost one or more electrons. Therefore it acquires a net negative or positive charge.
13 A chemical property of a substance that indicates an uneven distribution of charge within the molecule. A polar substance or drug mixes well with water but not with organic solvents and lipids. Polar or charged compounds do not cross cell membranes (lipid) very easily.